-
Solutions
Care Area Solutions
Standardize GCX mounting solutions throughout your healthcare facility.
IT and EMR Solutions
Mounting solutions for Information Technology and Electronic Medical Records equipment.
Mounting solutions based on your mounting location.
Medical Device Solutions
Mounting solutions designed specifically for your medical and patient monitoring devices.
-
Products

Arms
Variable Height

Carts and Roll Stands
Variable Height
Fixed Height
Accessories
- 10 x 25 mm Rails
- Barcode Scanner Mounts
- Baskets and Bins
- Cable Management
- Camera Mounts
- Channels
- CPU Mounts
- Data Capture Device Mounts
- Down Posts
- Drawers
- Gas Cylinder Mounts
- Gel Bottle Mount
- Handles
- Keyboard Trays and Plates
- Medical-Grade Power Strips
- Mountable Tablet Enclosures
- PolyQuip® Post and I.V. Bag Hanger Mounts
- Power Supply Mounts
- Probe Cup Mount
- Speaker Mount
- Tablet Integration
- Trays
- UPS Mounts
- USB Charging Modules
- VESA Adapter Plates
-
OEM Services
Mounting solutions for medical device OEM partners range from off-the-shelf to complete custom products.
Approach
Modalities
- Ventilation
- Ultrasound
- Patient Monitoring
- Telemedicine
- Surgical Navigation
- ECG Solutions
- Other Modalities
Our Expertise
-
Support
-
About GCX
Lilitab™ is now a part of GCX! Learn More
MD&M West 2026: See What’s New in Mounting and Mobility
Meet GCX at Booth #2343. Smarter OEM mounting for better clinical workflows. February 3-5, 2026. Anaheim Convention Center, CA
Why partner with GCX for OEM Services
If your team is evaluating mounting, mobility, and workflow integration for your next-generation device, GCX can help. We support OEM programs from early concept through production with design, testing, manufacturing, and global logistics.
For MD&M West 2026 attendees
Have a mounting challenge to solve in 2026? Meet our OEM team at MD&M West and get quick input on feasibility, safety, and next steps.
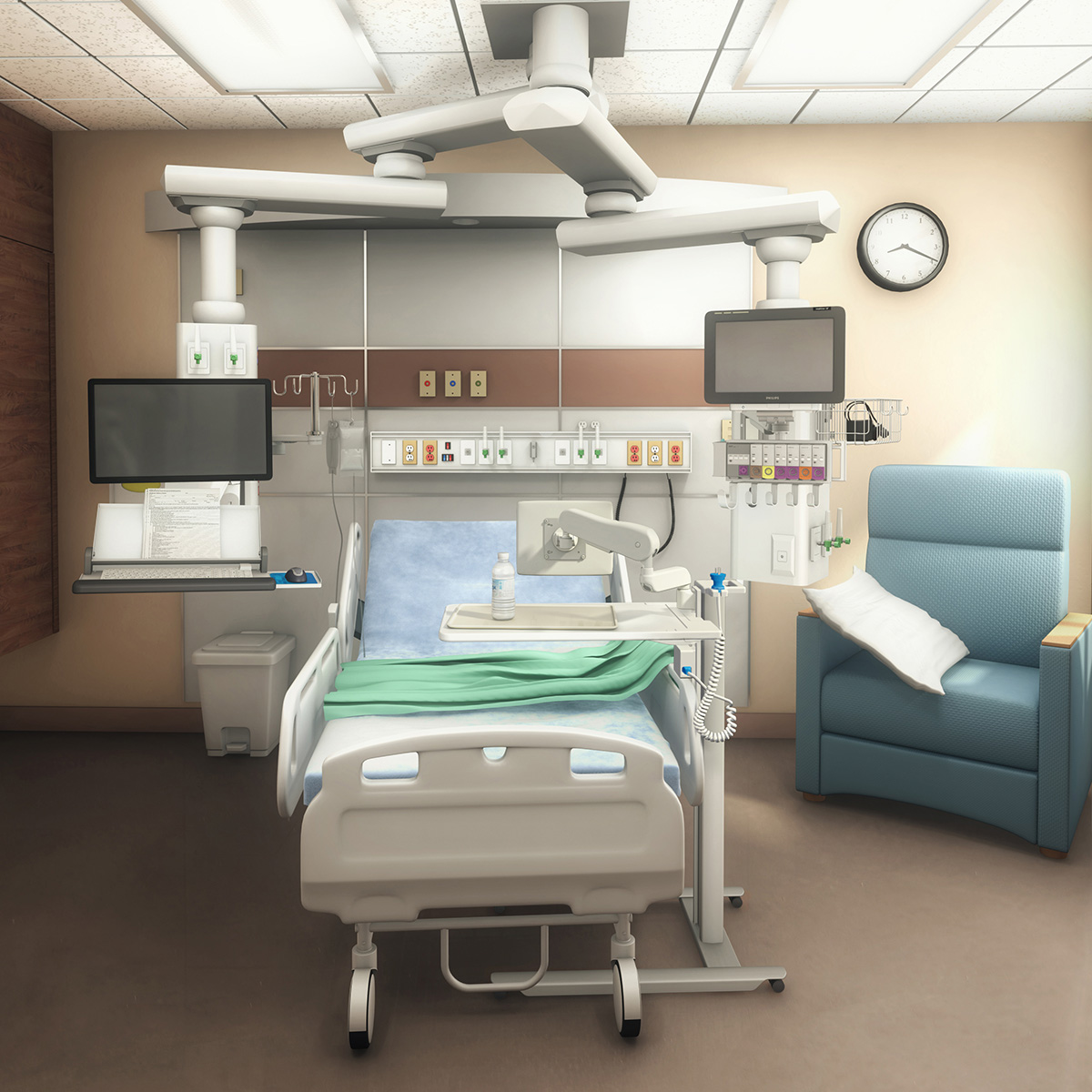
On-site discussion topics
- Choosing the right pathway: standard, custom, or hybrid to meet timeline and budget
- Designing for usability and aesthetics while supporting your device
- Validation approach and documentation considerations for IEC 60601-1 and related requirements
Let’s connect at MD&M West
Tell us what you are building, and we will follow up to schedule a booth meeting or a post-show OEM consult.
What you get with GCX
Off-the-shelf to fully custom
Build from proven standard components, create fully custom designs, or combine both into a hybrid solution.
Designed for healthcare environments
Improve ergonomics, cable management, cleanability, and caregiver access to your device in real-world workflows.
Quality and verification support
Designed and manufactured under ISO 13485 processes and tested to relevant standards such as IEC 60601-1.
OEM Service that scale with your program
Involve our engineering team early to create the right solution at the lowest cost. GCX helps get your device to market quickly with a full range of product development services.
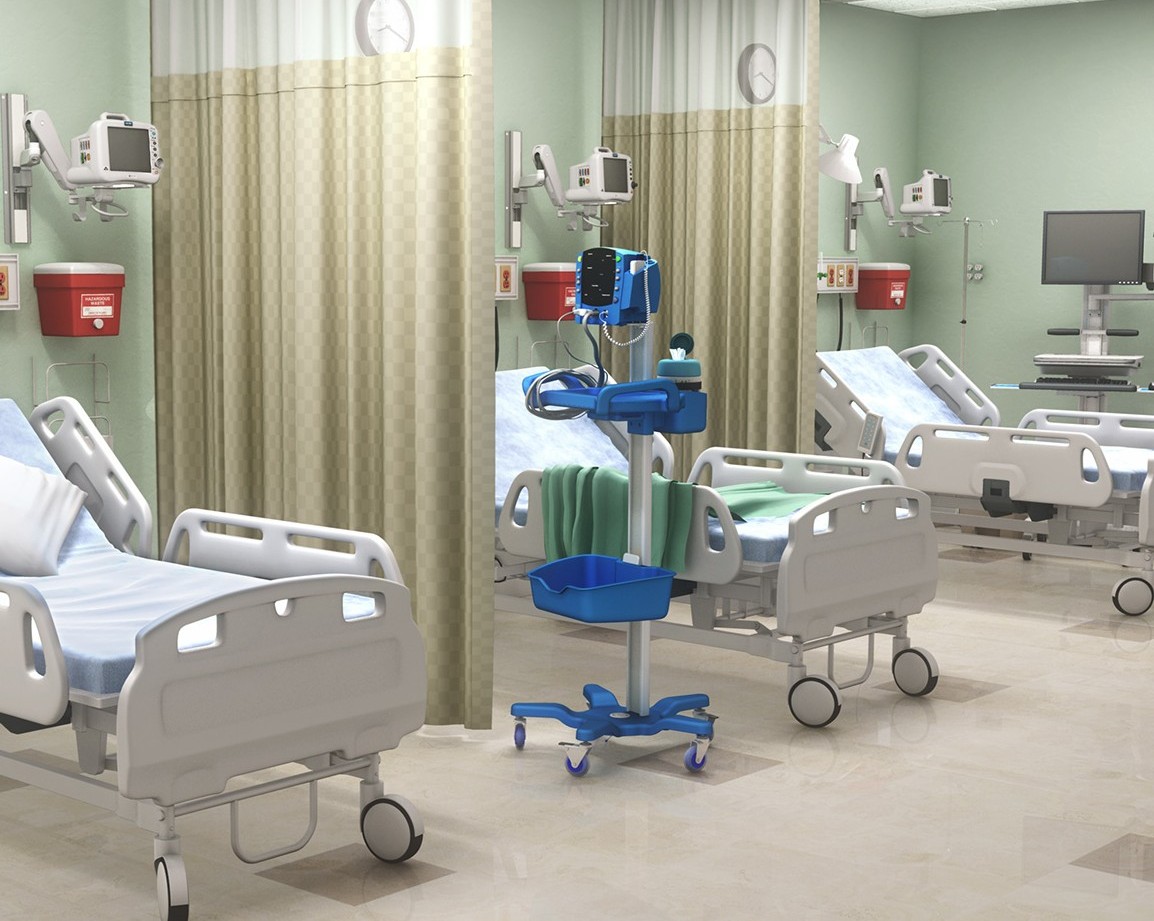
- Industrial design
- Mechanical design
- Rapid prototyping (in-house FDM and complex-surface machining)
- Engineering documentation
- Testing to recognized standards
- Manufacturing and tool design
- Technical writing
- Requirements development
- Label design
- Packaging design and testing
- Marketing materials
Built to support prototypes, ramp-up, and high-volume production
We help manage costs by matching changing production volumes to the appropriate manufacturing location. For prototypes, clinical trials, or low-quantity programs, GCX uses a highly flexible Northern California facility, then can transfer production to higher volume sites as quantities increase.

Manufacturing capabilities
- CNC milling and turning
- Sheet metal fabrication
- Aluminum extruding
- Injection molding
- Die casting
- Plastic forming
- Finishing
- Structural foam
Mounting solutions are tested through rigorous safety and reliability testing aligned to applicable industry standards, such as IEC 60601-1.
Where GCX OEM solutions show up
GCX expertise extends across many care areas and device categories, including: anesthesia, endoscopy, cardiology, neurology, IT equipment, patient monitoring, ventilation, ultrasound, urology, and more.
Do you offer off-the-shelf and custom solutions?
Yes. GCX supports standard products, fully custom designs, and hybrid solutions that combine standard and custom elements.
Can GCX help with prototyping and validation?
Yes. GCX offers rapid prototyping, engineering documentation, and testing to recognized standards, including IEC 60601-1 where applicable.
How do you support scaling production?
GCX supports programs from prototypes/clinical trials through higher-volume production and can support local assembly/distribution for drop ship or JIT replenishment.